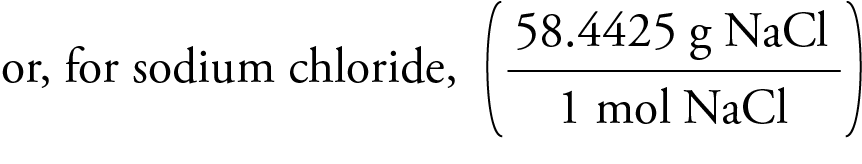
And don’t forget to put the unit g/mol to your final calculated molar mass. The first step to finding the molar mass of Sodium Carbonate is to count the number of each atom present in a single molecule using the chemical formula, Na2(CO3): Element Number of Atoms Na (Sodium) 2: C (Carbon) 1: O (Oxygen) 3: 2.First solve the brackets, then multiplications and at last do the final addition. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (NaBrO) Count i Weight i. Then, lookup atomic weights for each element in periodic table: Na: 22.98976928, Br: 79.904, O: 15.9994. Always follow the calculation order to avoid any mistakes in calculation. First, compute the number of each atom in NaBrO: Na: 1, Br: 1, O: 1.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole. :max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. Mass numbers of typical isotopes of Sodium are 23. I hope you have understood the short and simple calculation for finding the molar mass of NaCN. Sodium - Mass Number - Neutron Number - Na. Hence the Molar mass of NaCN is 49.008 g/mol. So, Molar mass of NaCN = Molar mass of 1 Sodium (Na) atom + Molar mass of 1 Carbon (C) atom + Molar mass of 1 Nitrogen (N) atom. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass): Molar Mass (g/mol) Na (Sodium) 22.98976928: C (Carbon) 12.0107 : H (Hydrogen) 1.00794: O (Oxygen) 15.9994: 3. You can see that in NaCN, there is 1 Sodium atom, 1 Carbon atom and 1 Nitrogen atom. In practice, the electrolysis process produces calcium metal as well but this is solidified in a collection pipe and returned back to the melt.Now, to calculate the molar mass of NaCN, you just have to add the molar mass of all the individual atoms that are present in NaCN. The electrolysis is carried out as a melt in a "Downs cell". However a mixture of NaCl (40%) and calcium chloride, CaCl 2 (60%) melts at about 580☌ and so much less energy and so expense is required for the electrolysis. Sodium chloride has a high melting point (> 800☌) meaning that it sould be expensive to melt it in order to carry out the electrolysis. Figure 3.4 Table salt, NaCl, contains an array of sodium and chloride ions combined in a 1:1 ratio. The formula mass for this compound is computed as 58.44 amu (see Figure 3.4 ). 23 Na is created in the carbon-burning process in stars by fusing two carbon atoms together this requires temperatures above 600 megakelvins and a star of at least three solar masses. It is easily recovered as a solid by drying. Sodium chloride is an ionic compound composed of sodium cations, Na +, and chloride anions, Cl, combined in a 1:1 ratio. Isotopes Twenty isotopes of sodium are known, but only 23 Na is stable. Now in NaOH, there is 1 Sodium atom, 1 Oxygen atom and 1 Hydrogen atom. Sodium is present as salt (sodium chloride, NaCl) in huge quantities in underground deposits (salt mines) and seawater and other natural waters. You can see the molar mass value of all the atoms from this periodic table. All syntheses require an electrolytic step as it is so difficult to add an electron to the poorly electronegative sodium ion Na +. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set). You can see that The molar mass of Sodium is 22.990 g/mol. So let’s look at the molar mass of Sodium, Carbon and Oxygen from the above periodic table. Now in Na2CO3, there are 2 Sodium atoms, 1 Carbon atom and 3 Oxygen atoms. Isolation: sodium would not normally be made in the laboratory as it is so readily available commercially. The molar mass of Na2O (Sodium Oxide) is: 61.977 grams/mol. You can see the molar mass value of all the atoms from this periodic table. The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
